
Estimates vaccine could be available for general public in January
Following last week’s authorization of the first COVID-19 vaccine set to be distributed in the U.S., Ochsner health system received 9,375 doses of the vaccine, this first shipment designated to inoculate thousands of its employees throughout the state.
Earlier this week, St. Charles Parish Hospital, managed by Ochsner Health, began vaccinating its frontline healthcare workers against COVID-19. Vaccinations are voluntary and in this first phase as outlined by the state and the CDC, only those employees with direct COVID-19 exposure are eligible. The vaccines are by Pfizer, and will require a second dose 21 days following the first shot for full protection.
The Pfizer vaccine is to first be designated for the hospital system’s employees considered to be most at risk of exposure to the virus. Shipments were to be made to three Ochsner facilities – in New Orleans, Lafayette and Shreveport – to be stored in “ultra cold” freezers at the facilities before being delivered to the system’s other hospitals throughout the state. Those freezers can store the vaccine at extremely low temperatures, approximately minus 70 degrees Celsius.
Ochsner is involved with 40 hospitals and 100 clinics around Louisiana and Mississippi, including St. Charles Parish Hospital in Luling.
Workers considered to be at highest risk of contracting COVID-19 would receive first priority for vaccination, officials said, as well as emergency room and urgent care facility workers. Priority is tiered by risk of exposure to the virus, not health conditions of individual employees.
Representatives said it’s anticipated that following health care workers, those living in nursing homes and long term care facilities would be next in line for priority. For the general public, it’s estimated the vaccine could be available sometime in January, but the official date has not been determined at this time.
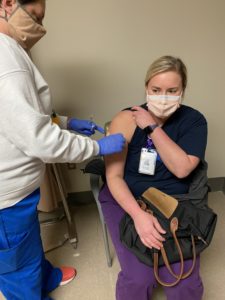
Ochsner is not requiring employees to be vaccinated, but chief medical officer Robert Hart said it would be encouraged.
“We are having lots of conversations about how important it is for all of us,” Hart said. “Waiting means more people in the hospital, more people dying, more people getting the disease … the only way we get out of this pandemic, because it’s too widespread in the community now to get out of this pandemic just by social mitigation efforts, the only way is herd immunity via the vaccine. And that requires all of us to be vaccinated.”
Hart said he would film himself being vaccinated to help further the effort to assure people of its safety.
Of the initial 9,375 doses, more than 5,000 were expected to arrive in the New Orleans area.
Doctors on the call stressed the safety of the vaccine and the detailed process in getting it ready for launch, and noted the side effects that have been reported from those who went through trials are byproducts of the body’s response to the vaccine, and that it is normal.
“Fortunately the vaccines are very safe, from the information we now have available from the trial,” said Dr. Katherine Baumgarten, Ochsner medical director of infection control and prevention. “The vaccine was used with over 30,000 participants and still showed safety (data) that was very impressive.
“The minor side effects include arm pain, a little bit of low grade fever and fatigue. Those are usually short lived, 24 to 48 hours after the vaccine is given.”
She said likewise, the timeline for release was shorter than for other vaccines historically because of a greater focus, not lack of care.
“No corners were cut in terms of safety. Many things were done at the same time with this vaccine. Instead of doing one thing for six months and another for a year, everything was done very quickly and brought to market at the same time,” Baumgarten said.





we are over 70 with medical problems. should be in 1C category. we are in Destrehan so please let us know when we can get the vaccine. thanks, John
Searching for a vaccine for my mom who at 76 years of age is still in the classroom teaching – She is a St. Charles Parish resident and teaches in a Jefferson Parish Catholic School – where can she go to get her vaccine????